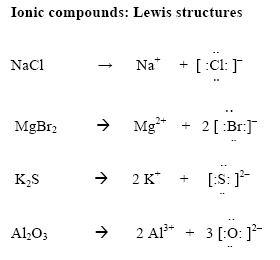FIRST LAB: Iron Nails and Copper Reaction
In this lab we tried to find the ratio of moles of copper formed to moles of iron reacted in the chemical reaction we did today for the lab.
Necessary 411:
When an iron nail is placed in a solution of Cupric Chloride the Iron will slowly dissolve. The dissolved iron will combine with chloride ions to form ferric chloride. In this activity you will determine the mass of iron consumed by comparing the mass of the nail before and after the reaction. Determine the number of moles of Iron this corresponds to. From this you will predict the number of moles of copper (ii) chloride that should be formed. By measuring the change in mass of the chloride solution in the beaker you can determine the mass of ferric chloride formed.
Procedure:
- Using a 10 mL graduated cylinder, measure 70 mL of Copper (ii) chloride
- Transfer the solution from the graduated cylinder to the 100ml beaker
- Record the mass of the beaker and solution
- Record the mass of the nail
- Place the nail in the beaker
- Wait between 2 and 5 minutes
- Remove the nail and record
Wear safety goggles and lab coats, as well as tie back and long hair and roll up any sleeves that are impeding your scientific process.
SECOND LAB: Molar Volume Lab
This experiment was designed as a way to test whether the molar volume of a gas at "around" STP is actually 22.4L
- In this experiment you need a lighter, a sink, a tap, a graduated cylinder and a scale
- First measure the mass of the lighter: record it
- Then fill your sink up with water and place your graduated cylinder upside down in it
- Then put your lighter underneath the cylinder and press the gas button
- As you press the gas button you will see little bubbles of coming from the lighter, trap these inside the cylinder
- Stop as your approach the 40mL mark
- Then dry off your lighter completely
- When your lighter is dry weight it again, and note the difference
This is the physical portion of the lab, for the rest of the lab you do theoretical speculations with the data you received and recorded whilst doing the experiment
- Next take the mass you got in the difference of your lighters and change that into moles( g -----mol/g)
- Now take your 40mL, convert it to L (.04L)
- Write a new equation for molar volume
- Volume over Mol
- 0.04L/Step1 = a number around 22.4
Careful with all your steps, and remember that because you're probable not operating at STP, your numbers WILL be a little bit off.